Tuesday, March 23, 2021
Yesterday, we discussed the legalities of off-label prescribing in pediatric anesthesia and how that climate is changing with regard to drug company advertising. Today, we’ll extend that discussion to a topic that’s recently been in our literature – the use of the supposedly “long-acting” local anesthetic called liposomal bupivacaine (Exparel).
Exparel was initially approved for surgical infiltration in 2011, but not as a “long-acting” preparation. More recently, it has been approved for use in brachial plexus block only. Notably, this was the first time the FDA granted an indication for a local anesthetic for a specific type of regional nerve block. They did so, probably in response to mounting criticism that the FDA was not doing enough to stem perioperative opioid use. But since its original approval, post-marketing studies on the effectiveness of Exparel have been mixed, at best.
Sandhu et al, performed a randomized and blinded (to the patient and study team but not the surgeon) study comparing plain bupivacaine and liposomal bupivacaine for “truncal” incisions, which included sternotomy, thoracotomy, minithoracotomy, and laparotomy in adults. A total of 140 patients per group were evaluated. The local anesthetic was injected into the tissue surrounding the incision at the time of closure. In brief, there were no differences in indices of postoperative pain control (e.g., pain scores, opioid use) or patient satisfaction up to and including postoperative day #3.
The February 2021 issue of Anesthesiology also contains some important updates on this effectiveness. In a systematic review and meta-analysis, Hussain et al. pooled the results from 9 trials that included 619 patients and concluded that “High-quality evidence does not support the use of perineural liposomal bupivacaine over nonliposomal bupivacaine for peripheral nerve blocks.” In the same issue, Ilfeld et al. published a comprehensive review of all studies that have examined the effectiveness of Exparel and concluded: “The preponderance of evidence fails to support the routine use of liposomal bupivacaine over standard local anesthetics.” These articles were accompanied by an editorial written by Dr. Mary Ellen McCann of Boston Children’s. Dr. McCann and I were on the same FDA advisory committee that evaluated the use of Exparel for regional nerve blocks. Even the studies the company submitted for approval showed marginal effectiveness, not only over plain bupivacaine, but even when compared with saline placebo. In her editorial, Dr. McCann writes:
“Once Exparel was approved, Pacira Biosciences began an aggressive and powerful marketing strategy. Between 2013 and 2019, they paid $25.8 million to more than 27,000 physicians for a variety of services including compensation for being a speaker or faculty at nonaccredited educational events. Sales of liposomal bupivacaine increased during this time with the company reporting a 25% growth in 2019 over 2018 with full-year revenues of $421 million in 2019. The cost of a single dose of 266 mg of Exparel brand liposomal bupivacaine is about $334. Nonliposomal bupivacaine costs about $3 per dose.”
My personal bias against using liposomal bupivacaine for regional nerve blocks is that there is no evidence (to my knowledge) that its accidental intravascular injection can be readily rescued by administration of intralipid. Administration of local anesthetic into a nerve sheath is much more likely to result in an accidental intravascular injection than indirect surgical infiltration into an open wound.
What’s the bottom line for us, as pediatric anesthesia practitioners? It seems to me that if liposomal bupivacaine was impressively better than plain bupivacaine, and worth its extra cost, we would know it by now, and its use would already by widespread in children. Until that happens, there is no reason to use it, and plenty of reasons to avoid it.
Medicolegal Monday, March 22, 2021
In today’s issue of Medicolegal Monday, we’ll take a break from tragic lawsuit cases in pediatric anesthesia to review an important legal aspect of our practice: off-label prescribing. As many of you know, pediatric anesthesia is one of the areas most affected by this practice. But, what actually is off-label prescribing? Is it legal? Do we put ourselves at medicolegal risk for this practice?
First, some background. When a pharmaceutical company has developed a novel medication that they wish to market for use in the U.S., they must first apply for permission to do so from the Food and Drug Administration (FDA). The process is complex, and can be confusing, but for those interested, I outlined the basic steps in an article in the October 2020 issue of Anesthesiology. The most basic requirements are for the company to demonstrate to the FDA that the drug is effective for a specific indication, and safe, when used at effective doses for that indication. This is usually accomplished by showing the FDA results from 2 separate, properly controlled trials. Once the FDA has approved the drug for sale, based on the indication set forth by the company, that indication is listed on the drug’s “label”, which most of us know as the “package insert”. By law, the company can then only market (i.e., advertise) that product for that specific indication (see Liu article for specific references to the laws). However, any licensed physician or dentist can use that drug for whatever purposes they want. When you use a drug for an indication that has not been approved by the FDA (eg, in children) it is called “off-label” use, and it is perfectly legal. Off-label prescribing is particularly common for pediatric uses, because of the underrepresentation of children in the clinical trials that led to approval.

Pediatric anesthesia is no exception. In a review published in A&A in 2012, Smith et al. demonstrated that out of 106 drugs potentially used in pediatric anesthesia or sedation, 36 (34%) were not FDA-labeled for use in any pediatric age group, and 30 (28%) were FDA-labeled for use in only specific age groups. We would like to believe that our off-label use of these drugs is justified because we know how to use them safely, and to omit them from our practice just because the FDA hasn’t approved it for that indication or population, would be worse than withholding their use. However, allowing drug companies to actively market a drug for an off-label use is an entirely different story. Historically, no one would argue that drug companies cannot be trusted to provide truthful information to clinicians. If they could, there would be no such thing as drug regulations (“If men were angels, no government would be necessary.” Federalist 51, Hamilton and Madison). As Liu et al. point out, “manufacturers have a financial incentive to oversell the benefits of their products and undersell the risks”.
This limitation on drug company marketing was the law of the land until 2012, when a Federal Appeals court in New York overturned it on grounds that it was unconstitutional (United States v. Caronia). The court reasoned that as long as the information was truthful and not misleading, the statute violated the 1st Amendment Freedom of Speech Clause.
The Liu paper cited above has analyzed the effects of that ruling on subsequent marketing and detailing by drug reps. In brief, they found that a little over half of 42 cases analyzed (both drug and device marketing) used the Caronia holding to influence their decisions against the FDA and for the drug or device companies, a scary trend indeed.
How do these new rulings that contradict those that were originally intended to protect patients influence our practice of pediatric anesthesia? It’s been years since I’ve talked with a pharmaceutical rep, since most hospitals no longer allow their presence. But we are still exposed to a barrage of ads in journals, websites, etc. on a daily basis. Plenty of research has shown that despite our belief otherwise, we are, indeed, susceptible to subconscious bias.
TRANSITIONS: Scott Markowitz, former CHOP fellow, now at Colorado Children’s will be the inaugural Vice-Chair for Professional Development and Diversity, Equity, and Inclusion in the Washington University Department of Anesthesiology…Dolores Njoku, my former pediatric intern at Rainbow Babies, previously at Johns Hopkins for her illustrious career, has been named the Division Chief of Pediatric Anesthesiology at Washington University…A double coup for Wash U! From CHOP, superstars John Fiadjoe and Jorge Galvez have taken new positions. John will be the Associate Chief of the Department of Anesthesiology, Critical Care, and Pain Medicine at Boston Children’s Hospital, and Jorge has been named Chief of Pediatric Anesthesiology at The Children’s Hospital and Medical Center in Omaha, Nebraska. Last but far from least, CHOP Uber-fellow, quadruple-boarded Andrew Renuart has accepted a position in the Department of Anesthesiology, Critical Care and Pain Medicine at Boston Children’s. With fellows like Andrew coming down the pike, our children and grandchildren are in great hands.
And on that note, as always on Mondays,
“Let’s be careful out there!”
Saturday, March 20, 2021

Good morning, and welcome to The Week in PAAD.
First and foremost, SHOUT-OUTS to PAAD’s newest Founding Member Subscribers: Lauren Lobaugh, Eric Holley, Micky Sofer, Maurine Heard, Tom Majcher, Lenny Naftilin, and Destiny Chau. THANK YOU! Your voluntary contributions to PAAD will be 100% donated to SPA’s Patient Safety, Education and Research Fund (PSERF) that supports junior investigators.
New PAAD additions: I’m a big fan of Politico’s Playbook, a daily email of current political events. Along with Heather Cox Richardson’s Letters from an American, it’s my morning coffee reading. Playbook has created a nice sense of community by including sections at the end of the newsletter with announcements of birthdays, job transitions, and other “mazel tovs”. Starting this Monday I’ll try to do the same with fellow PAADers. If you would like a shout-out, please send them to me at LitmanR@chop.edu and I’ll post them for the group (I already have 5 exciting transitions!).
ICYMI, here are the newsletters from the past week:
Medicolegal Monday March 15, 2021: Read about a tragic case of an easily preventable medication error in the OR.
Tuesday March 16, 2021: Is postoperative acute kidney injury in children a “thing”?
Wednesday March 17, 2021: Are opioids becoming obsolete in pediatric day surgery?
Throwback Thursday March 18, 2021: Distinguished Professor Myron Yaster discusses a classic paper on the origins of the 4-2-1 maintenance fluid formula. Take the IV fluid bag challenge.
TGI MH Friday, March 19, 2021: Should you do a non-triggering technique when a child’s extended family member may have had MH?
Hope you have a great weekend, and see you again Monday.
TGI MH Friday, March 19, 2021
Welcome to another issue of TGI MH Friday. Today we’ll discuss how to evaluate a very common problem we all face – the child with a vague history of MH in the extended family. But first, some new PAAD additions. I’m a big fan of Politico’s Playbook, a daily email of current political events. Along with Heather Cox Richardson’s Letters from an American, it’s my morning coffee reading. Playbook has created a nice sense of community by including sections at the end of the newsletter with announcements of birthdays, job transitions, and other “mazel tovs”. Starting Monday next week I’ll try to do the same with fellow PAADers. If you would like a shout-out, please send them to me at LitmanR@chop.edu and I’ll post them for the group.
Today’s case is one that we all encounter from time to time, and it comprises one of the most common calls we get on the MHAUS Hotline. An otherwise healthy 15-month-old boy presents for myringotomy and ear tube insertion for the first time. During the preoperative visit, the parents tell you that the mother’s aunt may have had an episode of MH as a child but was never formally tested for MH susceptibility. No one else in the family has ever had any problems with anesthesia, but those records are not available. Should this child now be considered MH susceptible and receive a non-triggering technique?
To me, there are several important issues to consider in this case: Based on the history, what is the chance that the child is truly MH susceptible? If you decide to go ahead with a non-triggering technique, how would you prepare the anesthesia machine? How would you then anesthetize the child? Finally, how would you counsel the family once the procedure is finished? Let’s take each of these in turn…
Unless we have convincing evidence to the contrary, we need to assume, for safety’s sake, that the aunt’s history of MH is real. How closely related does a “proband” have to be to warrant avoidance of MH triggering agents? Although a classic Mendelian inheritance of MH is an oversimplification, the autosomal dominant mode of inheritance is helpful to estimate the risk of MH susceptibility. Each offspring of an MH susceptible parent has a 50% risk to be MH susceptible. With every generation, the risk of MH susceptibility decreases by 50%. Therefore, the next generation (grandchildren) each have a risk of 25% and so on. If we assume that the child’s mother’s aunt inherited the MH mutation from one of her parents, it means that the child has a 12.5% chance of having inherited it. Using a conservatively high prevalence of MH variants of approximately 1 in 1,500 in the general population, it would take approximately 10 generations to decrease the calculated familial risk to be similar to that of the general population. What that means is that any familial history within 10 generations (essentially everyone) should be cause for suspicion of MH susceptibility and should prompt the use of a non-triggering general anesthetic.
Preparation of the anesthesia machine is relatively straightforward, as long as you have charcoal filters. Once they are attached to the inspiratory and expiratory limbs, clearing residual gas from the machine and anesthesia circuit only takes a few minutes, and there is usually no obstruction to OR throughput. Without the filters, flushing methods vary between anesthesia machines, and take much longer. For extra protection, we also either temporarily remove the vaporizers, or place a big piece of tape over them.

The non-triggering anesthesia technique is also straightforward, but not necessarily easy. There are many variations in practice. I would premedicate the child with oral midazolam, and then place an IV in the OR while the child breathes N2O. Occasionally, we’ll find an expert nurse who will place the IV in the preoperative holding area. Once the IV is placed, propofol and ketorolac can be administered for the procedure.
How, then, to counsel the family? If ever possible, the family member with the highest risk of MH susceptibility should be tested for MH in order to rule out, or confirm, the risk of MH in subsequent generations. That means going back to the maternal aunt and asking that person to get either a genetic test (confirms MH susceptibility), or a contracture biopsy (rules out MH susceptibility). In practice, neither of these get done in most of these cases, and the best one can do is help the parents understand the ramifications of an MH-susceptible label.
In the interest of brevity for this daily PAAD, many details of the above have been omitted, but complete information can be found on the MHAUS website.
As always, I hope everyone has a relaxing weekend, and special thanks to those on call. I hope your days and nights are quiet till Monday morning.
Throwback Thursday, March 18, 2021
In today’s edition of Throwback Thursday, I’m thrilled to include Dr. Myron Yaster as the first Guest Reviewer for PAAD. Myron needs no introduction. Most of you reading this newsletter will recognize him as one of the most well-respected and leading pediatric anesthesiologists in the history of our specialty. Today, he will review the origins of the 4-2-1 rule that has governed perioperative and hospital-based maintenance fluid therapy in children for decades.

The maintenance need for water in parenteral fluid therapy., HOLLIDAY MA, SEGAR WE. Pediatrics. 1957 May;19(5):823-32. PMID: 13431307
In this classic paper, Holliday and Segar proposed a simple-to-use formula that relates to the average caloric expenditure of a child of a given weight in kg:
- Infant up to 10 kg expend 100 kcal/kg;
- Children 10 – 20 kg expend 1000 kcal + 50 kcal/kg:
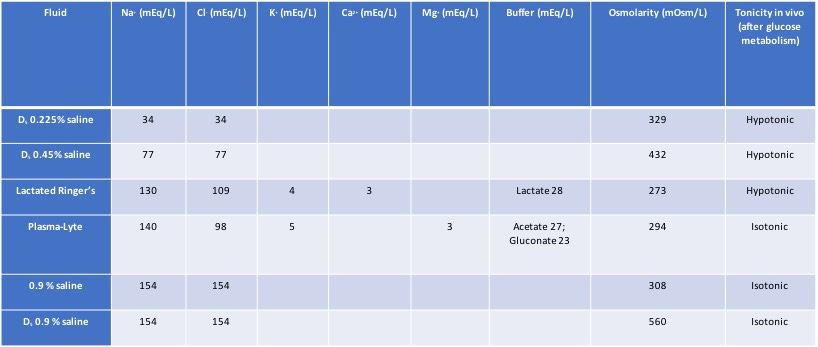
- Children > 20 kg expend 1500 kcal + 20 kcal/kg.The allowance of 50 mL/kg/24 hours will replace insensible losses and 66.7 ml/kg/24 hours will replace urinary losses so that 110 to 120 mL/100 kcal expended every 24 hours meets appropriate water maintenance needs. The math was further simplified as the “4-2-1 rule” and it is still used by anesthesiologists (and pediatricians) to this day. At the end of the article, Holliday and Segar conclude that daily electrolyte requirements for Na, Cl, and K are 3.0, 2.0, and 2.0 mEq/100 cal/day, respectively.
In a very important related and more recent paper, Ann Bailey and her colleagues point out “that these electrolyte requirements are theoretically met by the hypotonic maintenance fluid more commonly used in hospitalized children in the United States today, 5% dextrose with 0.2% normal saline. In Holliday and Segar’s original conclusions, it was emphasized that “these figures provide only maintenance needs for water. It is beyond the scope of this paper to consider repair of deficits or replacement of continuing abnormal losses of water.” Bailey adds, “Unfortunately, clinicians may often extrapolate the “4-2-1 rule” and the accompanying hypotonic solutions to clinical situations where they may not be appropriate and could, in fact, be harmful.”
Bailey et al. are spot on right! In the OR, we should ONLY be infusing isotonic solutions with Plasmalyte, Ringers Lactate, or (ab)normal saline. (I call it abnormal saline because there is nothing “normal” about it: it contains too much chloride and can produce a hyperchloremic metabolic acidosis). I believe we should always discontinue hypotonic solutions in the OR to avoid hyponatremia, a completely avoidable iatrogenic complication.
Because NPO times, particularly for liquids, continue to drop (it’s pretty clear it will or is already dropping to only an hour), and the risks of too much crystalloid are becoming increasingly clear, perhaps we need to rethink how much crystalloid we are infusing in the OR completely. We are almost certainly giving too much and in this era of ERAS, we probably should reconsider how we do this.
A last comment: Surprisingly, many of the trainees that I work with simply do not know the composition of most IV fluids. So, during the “boring” parts of an anesthetic I ask them to construct a table. This is what it should look like when done:
Pediatric Anesthesia Article of the Day
Announcing a new project! Every weekday I will review and comment on a recently published article relevant to the practice of pediatric anesthesia. It’s completely free. If you want it delivered to your inbox every morning, go here to subscribe: ronlitman.substack.com. But they will be posted here too – just click on the menu tab above.
New Pediatric Anesthesia Conference Announced: Save the Date, December 2, 2017
Great news! The Philadelphia Pediatric Anesthesia Consortium (PPAC) has been officially formed and consists of pediatric anesthesia representatives from Children’s Hospital of Philadelphia, Dupont Hospital for Children, St. Christopher’s Hospital for Children, and Jefferson Medical Center. PPAC is kicking off our beginnings with an inaugural conference on Saturday December 2, 2017 here at CHOP, with a theme of Cutting Edge and Controversial Topics in Pediatric Anesthesia. It will be an innovative Ted-style 1-day conference that targets new and controversial practices in pediatric anesthesia. Tentatively scheduled topics include anesthetic neurotoxicity in the developing infant, pain relief for Nuss procedures, management of anesthetic crises, malignant hyperthermia related diseases, best practices for hand-offs, and Dr. Alan Flake will tell us about his ground-breaking research in fetal physiology and viability, as featured this year in Nature. Stay tuned, more info to appear here as the lecture schedule is finalized.
When to Intubate during Neonatal Resuscitation?
I was catching up on some reading recently and came across this nice review of neonatal resuscitation in the April 22 issue of Lancet (Manley BJ, Owen LS, Hooper SB, et al. Towards evidence-based resuscitation of the newborn infant. The Lancet; 389(10079): 1639-48). It’s an excellent overview of the physiological process of birth and the principles of neonatal resuscitation. In the article, the authors reproduced the Newborn Life Support Algorithm of the UK Resuscitation Council. The choices within this algorithm rely on the appearance of “chest movement” during inflation. This term presumably means the rise of the thoracic cavity that is an indication of lung inflation. But in clinical practice, this is difficult to discern, especially in the newborn population, and is often confused with the movement associated with gastric inflation. Over the years, I have been asked to consult on several medical malpractice cases wherein a newborn suffered hypoxia despite the treating clinicians’ insistence that they were achieving adequate “chest movement”. These clinicians were so convinced of the adequacy of their bag-mask ventilation that they discounted the severely low oxyhemoglobin saturation and heart rate values, and did not consider the placement of an endotracheal tube or laryngeal mask airway. The adequacy of bag-mask ventilation should not be judged on “chest movement” but rather on achievement of normal oxyhemoglobin saturation and heart rate values. If these are subnormal, clinicians should presume that bag-mask ventilation is not sufficient, and the child should immediately receive tracheal intubation or placement of a laryngeal mask airway.
The most recent version (2015) of the AHA Neonatal Resuscitation has incorporated this decision into their treatment algorithm (https://doi.org/10.1161/CIR.0000000000000267. Circulation. 2015;132:S543-S560) and I’ve reproduced it here:
Christmas Eve
Hope everyone is having a restful Christmas Eve. It’s nearly 11 pm and the ORs here at CHOP are rockin’ with a swallowed penny, aspirated hot dog, and a first for me, bilateral emergency SCFE! Good thing we have great nursing and surgical colleagues, and way too much sugar everywhere.
MH Lecture at PGA, NYC, Tues Dec 16
I’ll be lecturing on MH at the PGA in NYC this Tuesday morning at 9 AM in a panel called “Recent Advances Affecting Your Practice”. The title of my talk is “Malignant Hyperthermia: Hot Off the Press!” Get it? It is scheduled to take place in the North Broadway Ballroom on the 6th Floor of the Marriott Marquis. I’ll be highlighting some recent changes in the way we diagnose and manage MH, including use of charcoal filters, and use of the new dantrolene preparation, Ryanodex. I will go over the types of patients that really need a non-triggering technique, and I’ll also discuss some recent changes to the MHAUS website, where we will be omitting recommendations for treatment of myoglobinuria, and the story behind our removal of the recommendation to avoid calcium channel blockers when also administering dantrolene. If you are there, make sure to stop and say hi.

